When people think of batteries today, one type usually comes to mind: the lithium-ion battery. It powers nearly everything, our phones, laptops, tools, and electric cars. Over the last three decades, lithium-ion has become the dominant energy storage technology, and for good reason. It is light, powerful, and rechargeable.
But lithium-ion wasn’t the first, and it certainly won’t be the last. Around the world, researchers are hard at work developing new battery chemistries that could be safer, cheaper, or better suited for specific tasks. Some of these technologies build on ideas that are centuries old, while others are cutting-edge innovations only made possible with modern science.
Let’s explore why the world needs alternatives to lithium and what some of the most promising new battery chemistries look like.
Why Look Beyond Lithium?
Lithium-ion batteries have changed the world, but they come with challenges:
- Safety risks – If punctured, overheated, or overcharged, lithium-ion cells can catch fire.
- Expensive materials – Metals like cobalt and nickel are costly and often mined under poor environmental and labor conditions.
- Environmental footprint – Manufacturing and recycling lithium-ion batteries produce significant carbon emissions.
- Scaling limits – As we move toward powering entire grids with renewable energy, lithium-ion may not be the best fit for large-scale storage.
As electric vehicles, solar panels, and wind farms become more common, we’ll need batteries that can store vast amounts of energy safely, cheaply, and sustainably. That’s where new chemistries come in.
Sodium-Based Batteries: Abundant and Affordable
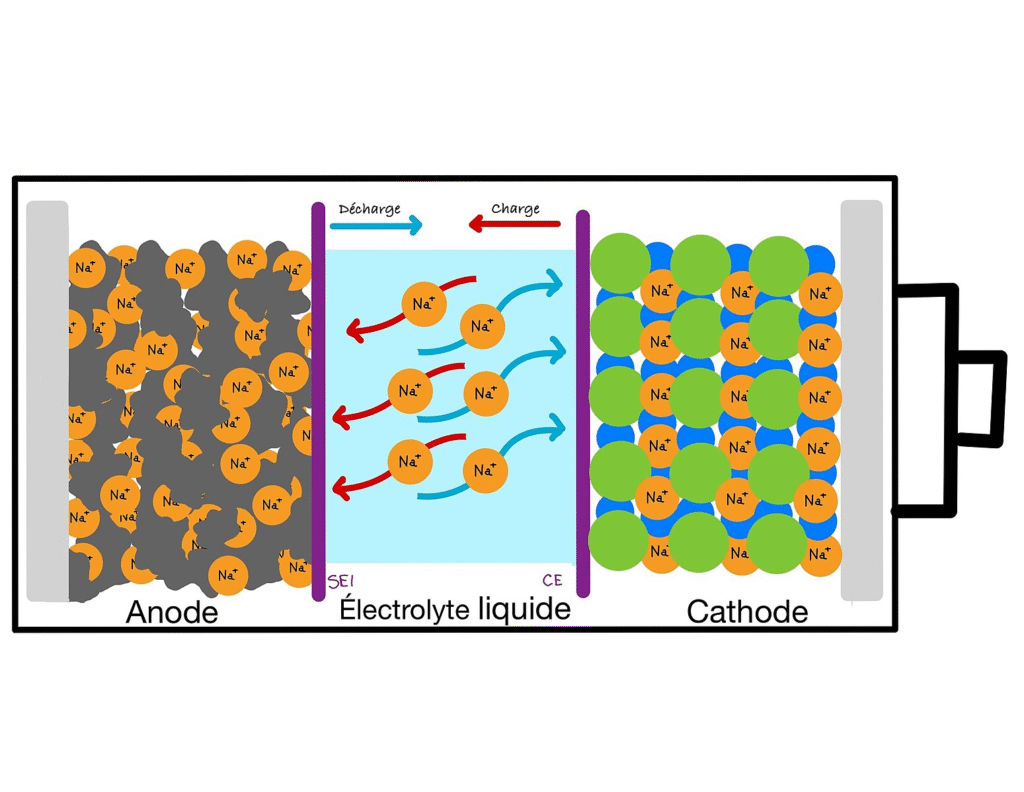
Sodium-ion batteries work in a similar way to lithium-ion, but instead of using lithium, they use sodium; the same element found in common table salt. Sodium is one of the most abundant elements on Earth, making it far cheaper to source.
While sodium-ion batteries generally store less energy per kilogram than lithium-ion, they have important advantages. They are less likely to overheat, making them safer, and their materials are widely available, which avoids the supply chain bottlenecks associated with lithium.
Sodium batteries are particularly promising for large-scale energy storage. Imagine a wind farm or solar power plant producing energy during the day. Instead of wasting excess electricity, it can be stored in huge sodium-ion battery banks and released later when demand rises. In these cases, size and weight are less important than cost and reliability.
Pros:
- Much cheaper raw materials
- Stable and less prone to overheating
- Ideal for grid storage where space is not a big concern
Cons:
- Lower energy density (meaning larger, heavier packs compared to lithium)
- Technology is still maturing compared to lithium
Several companies, especially in Asia, are already building pilot projects using sodium-ion batteries, proving that this technology is moving from theory to reality.
Magnesium and Calcium Batteries: More Charge in Less Space
Magnesium and calcium are exciting candidates because they can carry two positive charges per ion, compared to lithium’s one. In theory, this means a magnesium or calcium battery could store more energy in the same volume, offering higher energy density.
This would be a game-changer for electric vehicles, where space and weight are critical. A battery with twice the charge in the same footprint could extend the driving range without increasing vehicle size.
However, there are challenges. Both magnesium and calcium ions move more slowly than lithium ions, and they interact with electrolytes in ways that can cause unwanted chemical reactions. Designing stable and efficient electrolytes for these systems has proven difficult.
Despite the hurdles, research is moving quickly. Advances in materials science, especially in solid-state electrolytes, may soon unlock the potential of magnesium and calcium batteries.
Zinc and Aluminum Batteries: Safe, Recyclable, and Familiar
Zinc and aluminum are already widely used metals that are cheap, abundant, and easy to recycle. These qualities make them strong candidates for the next generation of sustainable batteries.
Zinc-air batteries use oxygen from the air as one of their reactants. This makes them lightweight, since the “fuel” doesn’t have to be stored inside the battery. They have already been used in small devices like hearing aids, but researchers are working to scale them up for bigger applications, such as renewable energy storage.
Aluminum-ion batteries have shown impressive results in laboratories, particularly in terms of fast charging. Some prototypes have recharged in just a few minutes. If this performance can be scaled, it could dramatically reduce EV charging times.
Because zinc and aluminum are so abundant, these batteries avoid many of the ethical and supply issues tied to lithium and cobalt. They may not completely replace lithium-ion, but they could complement it in applications where safety and recyclability matter most.
Molten Salt and Flow Batteries: Storing Energy for the Grid

Not all batteries need to be small or portable. Some are designed specifically for grid-scale storage—huge installations that hold energy for entire communities.
Molten salt batteries operate at high temperatures and use molten salt mixtures as electrolytes. They can store large amounts of energy and release it steadily over long periods, making them ideal for backup power.
Flow batteries use tanks of liquid electrolytes that are pumped through cells to generate electricity. The beauty of flow batteries is that their energy capacity can be increased simply by using bigger tanks, making them highly scalable.
Both of these designs are bulky, but that’s not a problem when they’re housed in large facilities near power plants or solar farms. They are less suited for cars or laptops, but they play an essential role in stabilizing renewable energy.
Fuel Cells: Electricity from Chemistry in Motion
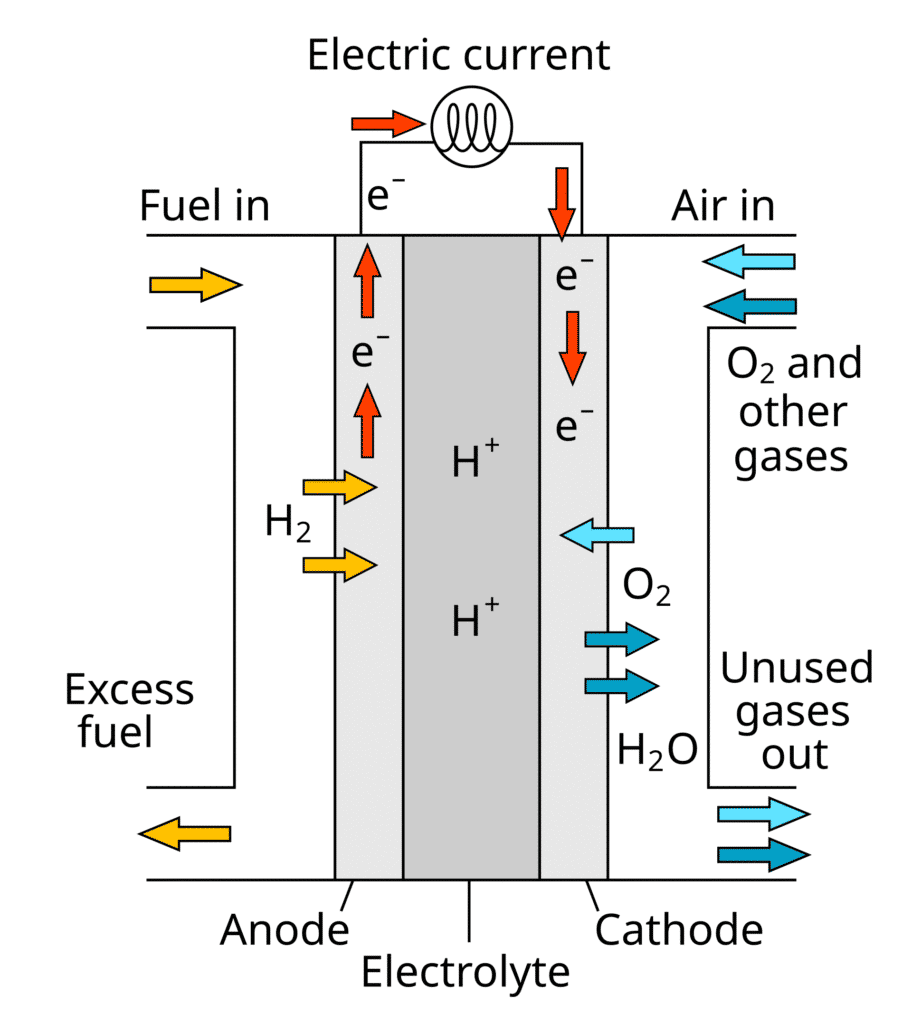
Fuel cells are often mentioned alongside batteries, but technically they are different. Instead of storing energy inside, a fuel cell produces electricity continuously as long as it has a fuel supply, such as hydrogen or methanol.
Fuel cells are already being tested in buses, trucks, and even aircraft. They offer the potential for long driving ranges and quick refueling compared to battery-electric vehicles. However, they depend on infrastructure for producing and delivering hydrogen, which is still under development in most regions.
For certain industries, like heavy transport and aviation, fuel cells could offer a cleaner alternative to fossil fuels.
The Future of Batteries: A Mix of Chemistries
The future of energy storage will not rely on a single battery chemistry. Instead, it will use a toolbox of solutions, each chosen for its unique advantages.
- Lithium-ion will likely remain the top choice for consumer electronics and many electric cars.
- Sodium-ion may dominate large-scale grid storage where low cost matters most.
- Magnesium and calcium could provide higher energy density for next-generation EVs.
- Zinc and aluminum offer safety and sustainability benefits.
- Molten salt and flow batteries will stabilize renewable energy on the grid.
- Fuel cells may power long-haul transport where batteries fall short.
By combining these technologies, we can build a flexible, resilient energy system.
References – Beyond Lithium
- Van den Bossche, A., & Moghaddam, A. F. (2021). Battery Management Systems and Inductive Balancing. The Institution of Engineering and Technology. (Overview of sodium, magnesium, calcium, zinc, aluminum, molten salt, fuel cells, and flow batteries).
- Tarascon, J.-M., & Armand, M. (2001). “Issues and challenges facing rechargeable lithium batteries.” Nature, 414(6861), 359–367.
- Dunn, B., Kamath, H., & Tarascon, J.-M. (2011). “Electrical energy storage for the grid: A battery of choices.” Science, 334(6058), 928–935.
- Images:
- Sodium-ion diagram: https://fr.wikipedia.org/wiki/Accumulateur_au_sodium
- Molten Salt: By RudolfSimon – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=18740786
- Fuel Cells: By R.Dervisoglu – Own work, based on http://en.wikipedia.org/wiki/File:Solid_oxide_fuel_cell.svg, Public Domain, https://commons.wikimedia.org/w/index.php?curid=19314043



